Warm Atomic Current: The Story of Radioisotope Thermoelectric Generators
From partisan kettles in WWII to Voyager probes 25 billion kilometers from Earth, radioisotope thermoelectric generators have been quietly powering civilization's most remote outposts for decades.
I wanted to write a short note about what an RTG is and where they live. But a short note didn't work out.
Pour yourself some coffee and get comfortable. Great physicists, cold snows, and distant space await us.
Don't pull the wires. Don't unscrew the bolts. Don't feed the bears.

We'll have to start our story from afar. This note will contain a (not so) little physics and a touch of history, but we won't dive too deep into the weeds. After that, there will be plenty of technology, good and varied. With photographs!
If you don't like physics (and you should) — scroll ahead to the atomic lighthouses. And if you don't like snow either — you can scroll even further, because there you'll find nothing less than outer space.
There's plenty to talk about here.
Physicists, an Experiment, and Two Metals
So, we should begin with the fact that the German physicist Thomas Johann Seebeck discovered the effect named after him in 1821.
If you build a closed electrical circuit from sequentially connected dissimilar conductors and provide a temperature difference by heating or cooling one of them, a current will flow in the circuit.
Such a current is called thermoelectric, and the resulting device is called a thermocouple.
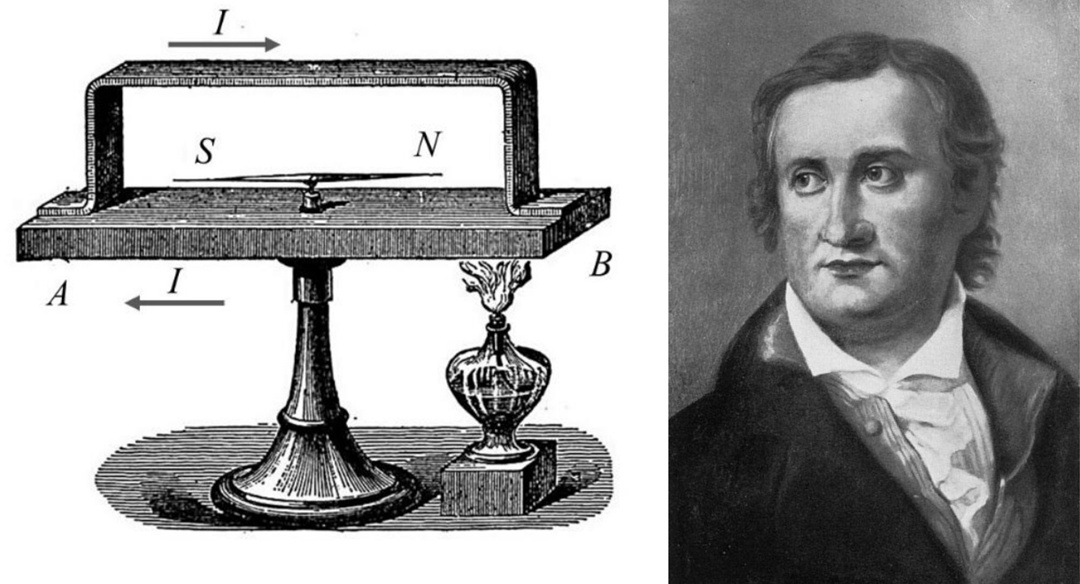
If the conductors are made of the same metal, no magic will happen — the electrical voltages between the ends of the thermocouple will simply cancel each other out.
Although nanoscale thermocouples exist that bypass this limitation in one interesting way, but that's a topic for another time.
Seebeck believed that under the influence of temperature differences, metals somehow became magnetized (some kind of "free magnetism" appeared), and electric current had nothing to do with it.
But the current arising in this process was later noticed by French physicist Jean Charles Peltier, whose name was given to the reverse effect.
True, he also didn't explain the physics of the process quite correctly — he linked it not to thermoelectricity at all, but to the hardness and electrical resistance of conductors. Peltier was very fond of the idea that heat is released differently in different conductors, so he happily applied it here too.
In the end, it was Russian physicist Emil Khristianovich Lenz who correctly understood what was actually happening. He conducted an experiment with two rods of bismuth and antimony — soldered them together, dripped water into the groove at the junction, and began running electric current back and forth. With one direction of current, the drop froze, and with the other — it thawed back.
At the junction of two different conductors (in a thermocouple it's called a junction), depending on the direction of current, heat can be either released or absorbed.
Quite the thermoelectric International we have here.
By the way, why did they think about it for so long? These scientists, really. It's a simple thing — take two different rods and connect them, then heat one end and cool the other and get current. Why did it take three whole physicists?
Well, the thing is that the simplicity is deceptive. The appearance of thermoelectricity (or more precisely, thermo-EMF — electromotive force) is a rather complex process.
Look — we already understood that heat is released at one contact of our thermocouple and absorbed by the other contact. Let's call it Peltier heat.
We mentioned Joule's law above.
When current passes through a conductor, part of it is converted into heat (the greater the resistance, the higher the heating).
This is also true for a thermocouple — voilà, now we know that Joule heat also arises in it.
And as if that weren't enough, inside each unevenly heated conductor, yet another form of heat is released, which is proportional to the rate of temperature change. Or absorbed, depending on the direction of current.
This phenomenon is called the Thomson effect (yes, another physicist, this time British — I told you, the whole world worked on thermoelectricity together). And so we now have Thomson heat.

There's as much different heat in this process as fur on a medium-sized cat — a whole lot.
So, the Seebeck, Peltier, and Thomson effects are to a certain degree interconnected — and all of them change sign depending on the direction of current, meaning they are reversible (they can heat, but they can also cool).
Joule's law says nothing about reversibility. It always only releases heat.
So what? We've now learned to convert thermal energy into electrical energy! Using a thermocouple! And that's no small thing!
Don't celebrate too early — those guys are about to strike back. Don't underestimate physics (or physicists).
The efficiency of such a scheme is very low — too small a fraction of thermal energy is converted to electrical energy.
And Joule heat is only half the trouble, although it also spoils things.
In metals, heat transfer from the hot contact to the cold one happens quite quickly in principle. Very quickly — their thermal conductivity is good (with rare exceptions), that's why they're metals.
But we need a temperature difference, remember? In these conditions, it disappears too quickly. That's why the efficiency of a metal thermocouple usually turns out to be... less than one percent.
Less than a percent?! Damn, that's completely useless.
As an indicator of some sort, maybe — to measure temperature. But for a generator, that kind of efficiency is just plain terrible.
How Radio Operators Boiled Water
But then Soviet physicists took up the cause.
It turned out that efficiency could be significantly increased by using semiconductors instead of metals in the thermocouple.
The thermogenerator based on semiconductor elements was originally developed by Yuri Petrovich Maslakovets, a colleague and student of Abram Fedorovich Ioffe.
Ioffe himself (a serious scientist — they don't name physics and technology institutes after just anyone) went on to excellently develop this topic in his monographs — "Energy Foundations of Thermoelectric Batteries from Semiconductors" and "Semiconductor Thermoelements," published in 1950 and 1956 respectively.

The use of semiconductors seriously increased thermocouple efficiency... which still remained low, at a maximum of five to ten percent.
But that's something we can work with.
During World War II, the laboratory for thermoelectric properties of semiconductors, headed by Maslakovets (in 1941 it was evacuated to Kazan along with the Leningrad Physics and Technology Institute), worked on creating thermoelectric power sources for partisan and sabotage units.
The result was the thermoelectric generator TG-1 — the "Ioffe kettle."
Several dozen thermocouples made of zinc antimonide (SbZn) and constantan (a copper-based alloy with additions of nickel and manganese) were connected in a circuit.
They were immersed in a water-filled vessel placed on a campfire. The bottom of the kettle heated up to 250-300°C, touching the "hot" parts of the thermocouples. And boiling water, as we know, has a temperature of 100°C, which (surprise!) cooled the "cold" parts.
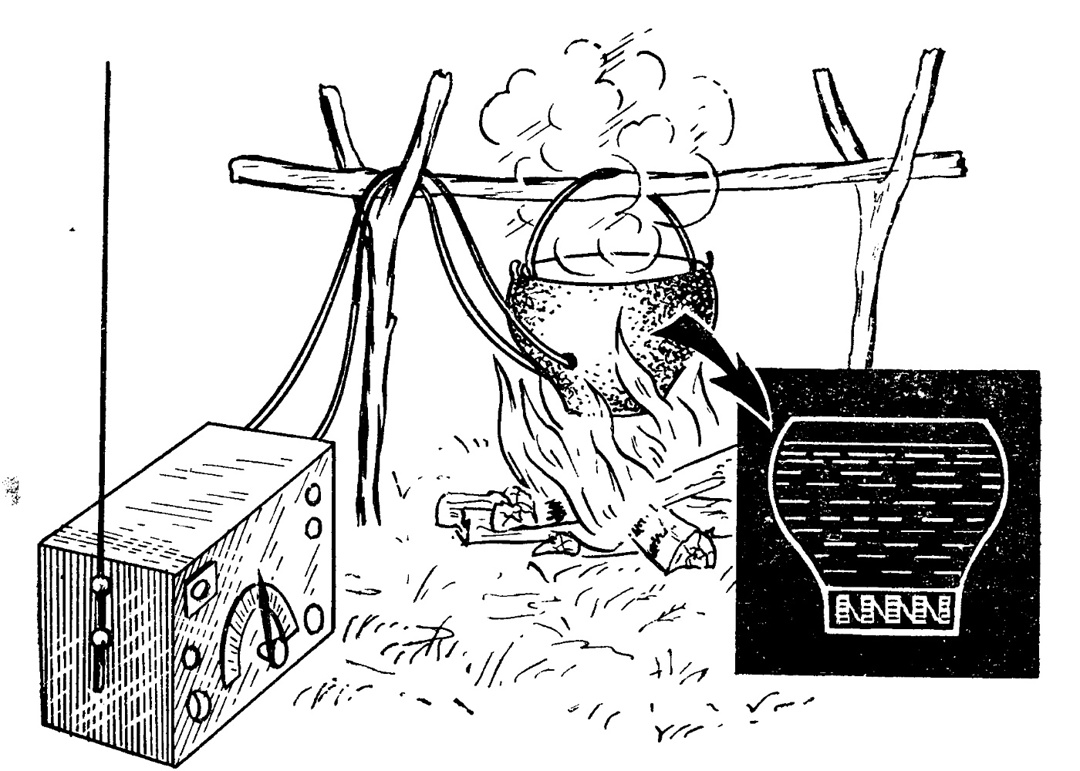
The device's power was modest, but sufficient to charge batteries for a portable radio station.
In peacetime, work on thermogenerators continued. Several devices running on charcoal were created, with air or water cooling (essentially, these were something like small stoves with a thermogenerator attached to the firebox).
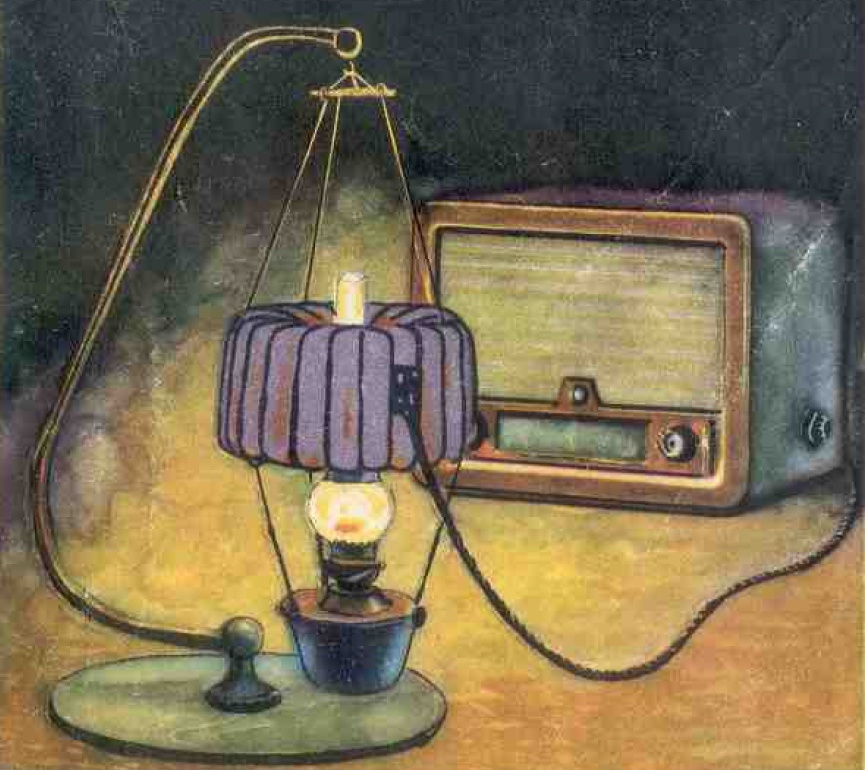
But it's more convenient to use not coal, but liquid fuel — for example, kerosene.
And here we can combine the useful with the pleasant — let the heat source for our thermogenerator be a kerosene lamp! At the time, in places that still had problems with electricity, rooms were lit by kerosene lamps — and since that was the case, a thermogenerator that could be placed right on top of one would be very convenient.
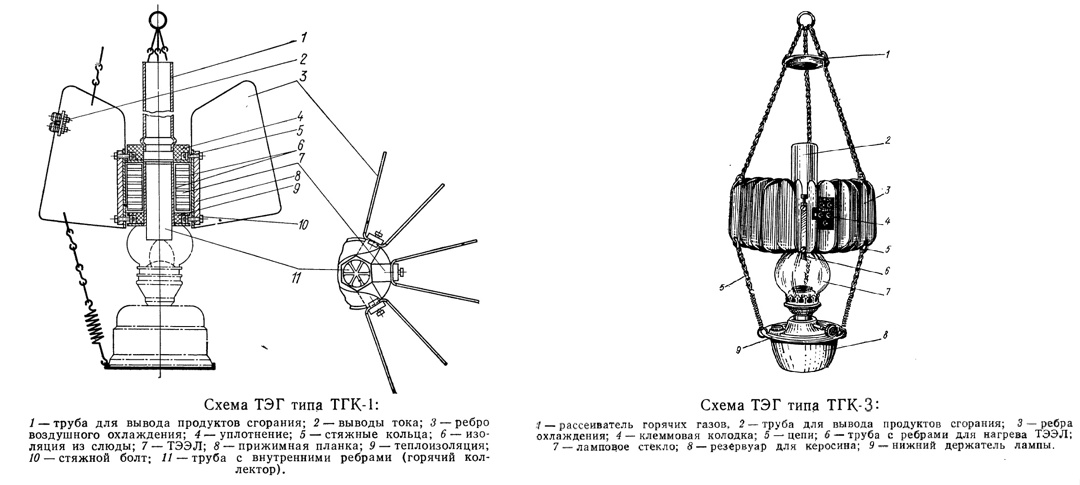
Light the kerosene lamp in the evening and switch on the radio at the same time. Wonderful!
It was precisely the TGK-1 or its descendant TGK-3 (which stands for kerosene thermogenerator with 3-watt output) that some of you might have encountered in real life. At least as a curiosity sitting on a far shelf.

In their time, the TGK generators played an important role in bringing radio to rural areas — until full-fledged electrical power arrived there.
Canned Heat
Well, alright. So we've learned to simply and reliably, if not too efficiently, generate electrical current. We even tested the technology in real applications.
And simplicity and reliability are good things — engineers love that. The device could come in handy somewhere else... But you can't exactly light a campfire everywhere. Or even burn kerosene — still not quite suitable.
And charging a radio is nice, of course, but we'd like something more serious too.
If we want to provide enough power for serious equipment (even if it's not the most power-hungry kind) this way, then the temperature difference across the ends of our thermocouple needs to be quite large, several hundred degrees Celsius.
Where can we get that without burning fuel? Where do we find a convenient heat source that works long enough and reliably enough?
The friendly peaceful atom comes to the rescue (brother of the mean military atom).
So, now we'll make a small nuclear reactor? No.
The trouble is that a reactor will never be truly small, and it also needs constant monitoring. We've decided that we won't leave personnel near the installation — especially since a nuclear technology engineer is a rare bird, you can't put one at every lighthouse.
Actually, small reactors do exist. They can even be unattended (also a topic for a separate long conversation). But more on that later.
And still, a nuclear reactor is complicated.
But there's another way to get heat from nuclear fuel — to use the energy released during its natural radioactive decay.
Let's simply place nuclear fuel in a sealed capsule and use the heat it releases on its own.
The resulting device will be structurally much simpler and more reliable than a reactor. And it will be autonomous in operation, requiring no maintenance at all.
It doesn't need to be monitored, since no chain reaction occurs inside. But heat generation happens constantly — until, after a certain time, the fuel element degrades completely, ceasing to maintain the temperature we need.

By the way, what fuel will we choose? There's a dilemma here — we want both more heat and a longer half-life so the device works long and efficiently.
Originally, I wanted to simply write that improving (increasing) one parameter inevitably worsens the other — well, if nuclear fuel releases a lot of heat, then its half-life is too short.
On one hand — it sounds logical enough. The Geiger-Nuttall law and all that. But on the other hand — this is a massive oversimplification, bordering on incorrect information.
But things aren't that simple. Take, for example, uranium-232 with a half-life of nearly 70 years and compare it with strontium-90, whose half-life is less than 30 years.
You'd think strontium-90 should release twice as much energy?
No, because uranium-232 has a very long decay chain with nuclides that emit hard gamma rays. It both decays longer and releases far more energy. Sorry, strontium-90, but you don't stand a chance here.
To simplify, from the substances available to us we can choose between strontium-90 (half-life of 29 years, initially releasing about 0.45 W of thermal energy per gram) and plutonium-238 (half-life of 88 years and 0.54 W of thermal energy per gram respectively).
So, plutonium is excellent, but it's far more expensive (let's save it for special cases when the device needs to run as long as possible), so as fuel we'll take strontium-90. We place it in our capsule.
Now let's combine the thermoelectric converter (since we've decided to work with it) and the radioisotope heat source through a thermal junction, place them in a thick housing with radiation shielding, and hermetically seal the lid.
All that remains is to mount a radiator on this housing for heat dissipation (remember, we need that temperature difference), connect wires to the terminals — and voilà, we get a radioisotope thermoelectric generator, or RTG for short.
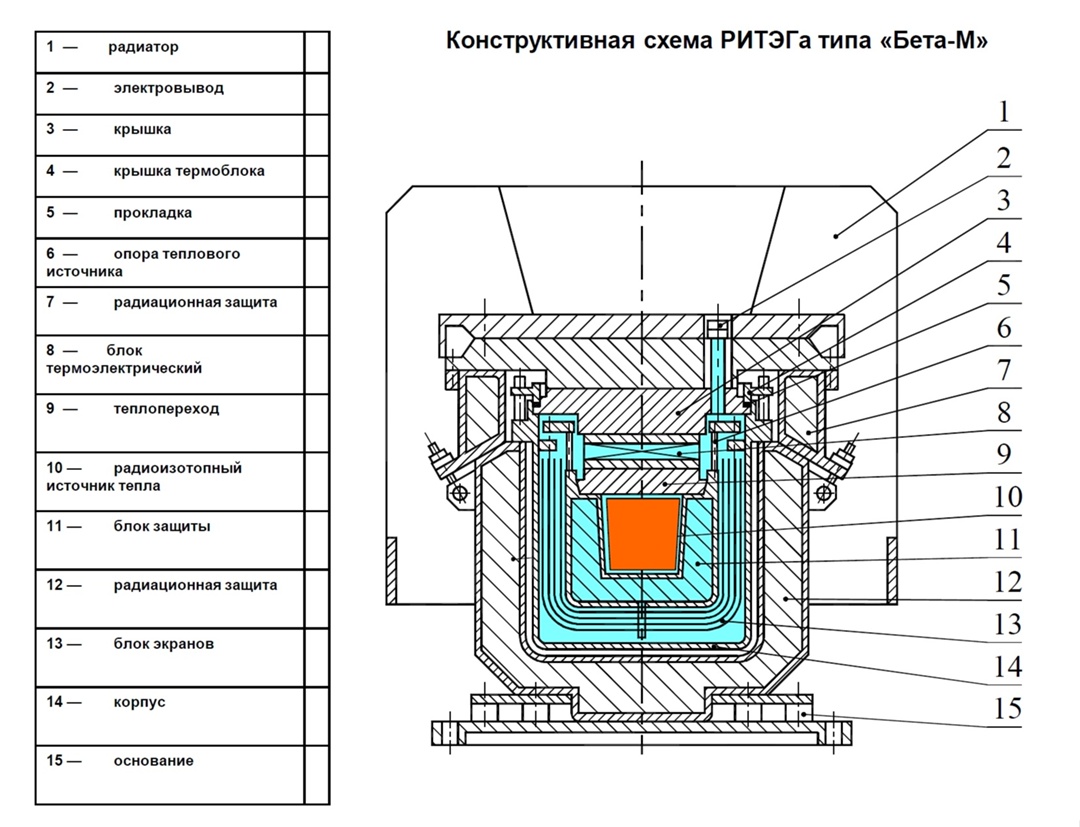
Here it is, the hero of our article, finally. With RTGs, things will get much more interesting.
Light in the Polar Night
Let's be honest — there are places on Earth where leaving people for long periods is inhumane and too expensive. But sometimes equipment needs to be placed there — for example, a lighthouse for passing ships. Or even a radio beacon. Or a weather station.
The equipment can operate automatically, but it needs electric current. And here a problem arises.
Running a power line across difficult terrain and building a transformer substation in the middle of nowhere for a lone lighthouse seems like overkill. Human settlements may be hundreds of impassable kilometers from the site, which means diesel generators are out. And wind generators too — due to their moving parts, they require periodic maintenance (though not too frequent).
Solar panels? That's trendy and eco-friendly, but in the north there are periodic polar nights when they'll be useless. Install a battery bank? Alas, they suffer terribly in the cold, and the polar night is far too long.
But now we have the RTG!
We need to set up a lighthouse somewhere on Novaya Zemlya among the snows and rocks? No attendant needed — let the equipment work automatically, and for power supply we place a brand-new RTG in the shed next door, run some wires — and it delivers sufficient power for the instruments to operate for the entire warranty period (ten years), demanding absolutely nothing in return.
"Just run some wires" is, of course, quite a simplification — most navigation equipment operates in pulse mode, so for such installations the RTG comes bundled not only with connecting cables but also with energy storage units (battery packs).

Moreover, during operation it turned out that an RTG running on strontium isotopes can last even fifteen years. And even twenty-five, if pressed (though by then it outputs only half the power — don't forget about half-life).
What a celebration! The device has absolutely no moving parts, which means no rapid wear from friction and vibration (maintaining something where nothing rotates is needed far less often, take it from a technician) — the latter also allows its use to power vibration-sensitive seismic equipment. RTGs also don't create radio interference that could disturb other equipment.
It doesn't require any serious maintenance either, since the capsule is hermetically sealed. Because the housing needs to be robust and massive anyway for adequate radiation shielding, RTGs are also resistant to harsh weather — many of its modifications were designed to operate outdoors (the main thing is to prepare a sufficiently level platform and fence it off).

That's why the USSR cranked out over a thousand RTGs of various models. The majority (about 850 units) went to support maritime or river navigation (mainly to the Northern Sea Route, into harsh conditions). 50 units were sent to automatic weather stations (and to polar stations in Antarctica). They proved useful at radio relay stations for long-range communications, and the military took them for their needs too.

About one hundred fifty to two hundred such installations were also built in the US — strictly speaking, the first RTG was developed there in 1954. They were used for the same purposes — navigation, powering Arctic equipment and radar systems in harsh conditions somewhere in Alaska, or military needs.


By the way. An RTG looks fairly simple in its operating principle — even simpler than a diesel engine. So why didn't everyone start making them? Toss some strontium in a barrel, stick in a thermocouple, run some wires — and you've got a battery for a couple of decades!
The obvious problem is the nuclear fuel itself, whose cost accounts for about 90% of the RTG's price. The aforementioned strontium-90 and plutonium-238 are (as a rule) produced in nuclear reactors, which already significantly narrows the number of contenders who can obtain them.
Furthermore, simplicity of design doesn't guarantee simplicity of manufacture.
The RTG housing uses depleted uranium for radiation shielding (it blocks radioactivity well due to its high atomic weight). The casing must be extremely robust to prevent breach of the radioisotope source.
And the thermoelectric converter needs to be made as high-quality as possible so its reliability lasts the device's entire service life (remember, we're talking about decades).
The thermoelectric converter is used here as the main example — also because the majority of Soviet RTGs for "terrestrial" applications were built with it. But in general, you can attach a thermionic converter, or a thermophotovoltaic element, or a Stirling engine like the American ASRG (see below) to such a convenient heat source. Where there's a will, there's a way.

And so hundreds of lighthouses, radio beacons, weather stations, and other far-flung things were powered by our trusty RTGs.
They do have their downsides, of course — electrical power output is modest because the efficiency of the thermoelectric installation is very low.
Yes, the electrical power of an RTG, depending on the model, is only tens of watts. But for the navigation equipment we're interested in, that's enough.
All That Glitters Is Not Gold (Sometimes It's Strontium)
When you laboriously drag a sealed barrel of nuclear fuel with wires sticking out of it to the edge of the world — you probably don't even imagine that someone in the world would actually walk all the way out here. At least not voluntarily, rather than by duty.
But such people turned up.
With the collapse of the Soviet Union, control over some of the lighthouses along the Northern Sea Route weakened. Though even before that, the most remote sites were visited for maintenance only once a year, or even less often.
Sometimes this depends on whether there's a bear currently roaming the lighthouse island.
I'm not joking. I came across an interview with someone who works at the Hydrographic Enterprise that services lighthouses (the navigation equipment department) — they visit lighthouses by boat during navigation season, roughly once a year. They check if everything's in order, change light bulbs, and so on.
Well, sometimes it turned out that bears were on the lighthouse islands. They'd arrive in late autumn or winter, walking across the ice. And once the ice melted, they couldn't leave the lighthouse anymore.
In such cases, the brave repair crew... sailed away from the lighthouse and prepared a report that maintenance of the given site was not possible this time due to dangerous fauna.
They'd return to the site the following year. The equipment generally permitted such liberties in maintenance — after all, it's automatic.
And so the orphaned lighthouses were soon being visited by dubious characters in pursuit of scrap metal.
The looters' logic was simple — if something mysterious sits on the site's grounds, it must surely be valuable (and in a way, they were right). From some RTGs they simply cut the electrical cables (also not good — idle mode is undesirable for them), while others they broke open (unscrewing connection bolts or trying to cut through the thick metal with gas torches).
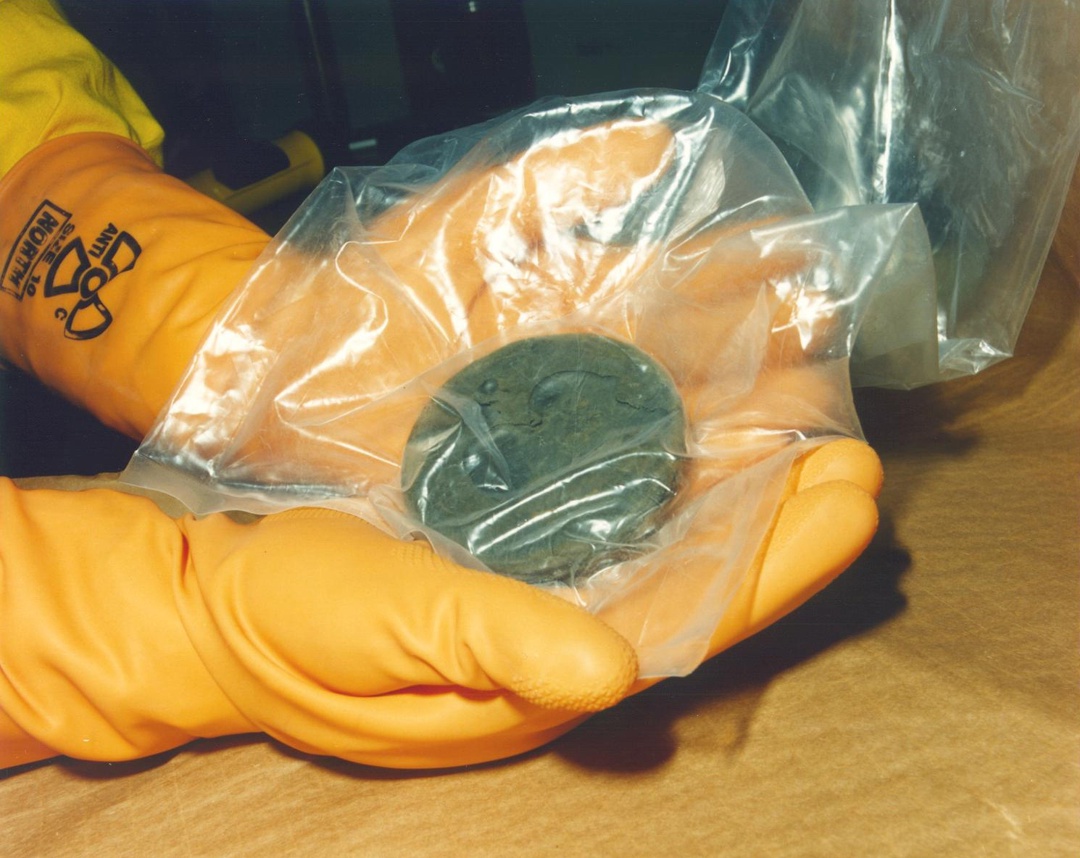
We already know that inside the device there's a capsule with nuclear fuel that emits ferocious radiation. But for some scrap metal hunters, this came as a surprise.
Well, I'd like to believe they simply didn't know they were cracking open something that radiates horribly.
Although radiation only kills those who believe in it, right?
As a rule, the radioactive elements were simply discarded on the spot — most likely, this didn't pass without consequences for the health of those who discarded them.
Moreover, back in 1983, one unpleasant incident occurred. In Chukotka, one RTG was seriously damaged in transit to its installation site — and the personnel concealed this fact from management, hoping that at such a remote site, it wouldn't be noticed for a while.
And indeed that's what happened — the damage was discovered by a Gosatomnadzor commission, but only in 1997.
The human factor is ineradicable, what can you say.
In the late 1990s, the unenviable state of RTGs in post-Soviet territory caught the attention of the IAEA — the International Atomic Energy Agency, a UN system organization with broad authority over regulating the peaceful (and not-so-peaceful) use of atomic energy.
IAEA personnel were extremely surprised when they learned that some RTGs in the 1990s had simply been abandoned when the organizations responsible for maintaining them fell into disrepair.
Gentlemen, we're doing everything we can to enforce the nuclear non-proliferation regime, and you've got strontium capsules scattered all over the North?! What if some very bad people find them and make a very, very bad dirty bomb? Have you thought about that?
And actually, a radiation leak can happen without people involved, you know — you've abandoned RTGs God knows how and God knows where, who knows what happens there, from earthquakes to manufacturing defects!
In short, stop this immediately, let's get rid of these things!
In fairness, the US was also winding down RTGs in remote areas around the same time. For roughly the same reasons — in the name of ecology and safety.
So in the early 2000s, within the framework of an international IAEA program for RTG disposal, a titanic effort to find them began. Information about the locations of sites was gathered bit by bit, and most of the devices could only be evacuated by helicopter.
In the chaos of the 1990s, much documentation was lost — for example, there was a case where a factory simply couldn't answer the question of how many RTGs it had even produced.

In the end, out of over a thousand RTGs, nearly all were sent to storage or disassembly.
Two were never found — and their fate is ambiguous. One (Shmidtovskiy district, Chukotka, Kyvekwyn River) is believed to have been buried in sand as a result of a severe storm; the other was dropped into the Sea of Okhotsk back in 1987 during helicopter transport. Both are still being actively searched for.
In total, the USSR created 1,007 land-based RTGs — not counting those that flew into space (oops, another small spoiler for the second half of this article).
But the population data comes from the IAEA itself, and as mentioned above, it was gathered with great difficulty.
So... well, they might have forgotten about a dozen or two, these things happen. Especially since some RTGs were in military hands — and they keep their secrets secret. Meanwhile, journalists from Komsomolskaya Pravda have a theory that 39 RTGs belonging to the Pacific Fleet were quietly dumped at sea. And reportedly, it's not even certain that the naval brass still remember where exactly they did their dirty deed. But if even the IAEA had no questions about the navy's RTGs... well, let's just say everything turned out fine with them in the end.
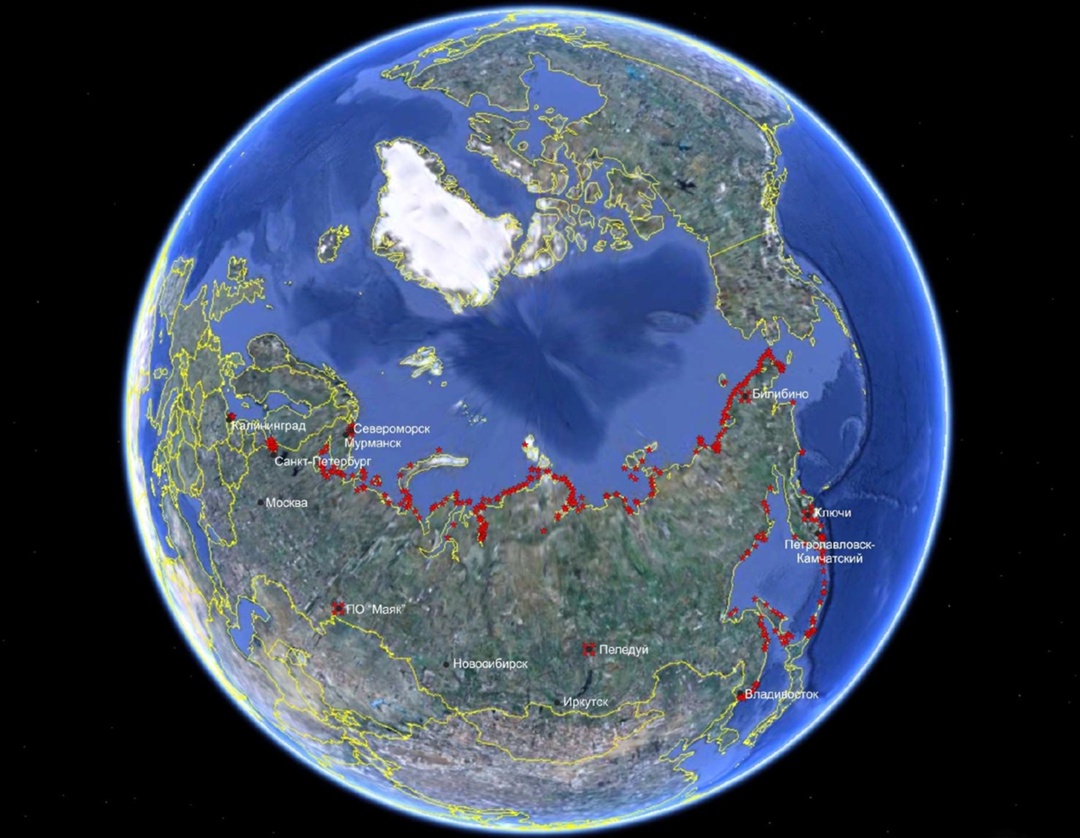
The lighthouses of the Northern Sea Route and other facilities in hard-to-reach places were gradually switched to alternative power sources. This work was handled by the Kurchatov Institute — and the task proved difficult.
In the end, photovoltaic power systems were used as replacements, consisting of solar panels, powerful battery banks, and a sophisticated automation system responsible for power supply reliability and transmitting the necessary data to the service organization.
But some problems remain relevant — the further north the lighthouse, the worse things are for solar panel performance and the longer the polar night. In harsh conditions, batteries (even specially prepared ones) degrade too quickly. Solar panels can be supplemented by wind generators, but then the installation needs frequent maintenance.
Unfortunately, to this day there is no power source matching the reliability and operational lifespan of RTGs.


The Loneliest Battery in the Universe
Let's go somewhere further... Actually, this is exactly where it all started for RTGs.
Originally, they were developed for spacecraft. RTGs have been widely used there ever since — and quite successfully. Today, the expanses of space are their primary habitat.

Although when it comes to generating electricity in space, the first thing that comes to mind is solar panels. Spread them out in space and fly along happily while the Sun does all the work for you!
You'd think that in space, solar panels wouldn't be hindered by weather or the change of day and night.
Ideal conditions!
All our lives we've heard that you can't run from your problems forever, but it turns out you can — you just needed to run at escape velocity and away from Earth.

But as the joke goes, there's a nuance — the Solar System turns out to be not all that solar.
It depends on where exactly we're flying.
If we're heading somewhere toward Jupiter or even farther from the Sun (Uranus, Neptune, Pluto, and onward toward the Oort Cloud) — solar energy may no longer be enough for us.
In physics, there's this thing called the inverse-square law — what matters for us is that according to it, the intensity of solar radiation decreases proportionally to the square of the distance from the Sun.
For example, from the Sun to Earth is about 149 million kilometers (this distance is called an astronomical unit, AU), while to Jupiter it's already 778 million kilometers, meaning five times farther.
That means solar panels in Jupiter's orbit would produce twenty-five times less electricity than in Earth's orbit.
Strictly speaking, planets orbit the Sun in elliptical orbits — thus the distance from Earth to the Sun is not constant.
Throughout the year it varies from a minimum of 147 million kilometers (when Earth is said to be at perihelion) to a maximum of 152 million kilometers (at aphelion).
But for our purposes right now this doesn't matter much — the difference with Jupiter is obvious either way.
Although progress doesn't stand still, and there's already talk that modern solar panels (with all sorts of tricks like solar concentrators with coolant circulation) could allow spacecraft to operate normally even near Jupiter.
But still, the farther from the Sun we fly — the sadder it gets. For a spacecraft, we need to generate a certain amount of power, and solar panels in that case need to be made larger and larger.
And a large solar panel area for a spacecraft means what? Exactly — during launch they'll need to be kept folded, and deployed somehow later.
The trouble is that something pre-folded may simply fail to deploy at the crucial moment. And it's one thing if such panels are meant for an inhabited station with specially trained astronauts — but what if the problem occurs on an automated probe that no one in space can help?
And such cases have already occurred — for example, with NASA's Lucy probe in 2021, when one of the solar panels didn't fully deploy because the drive cable got tangled on the spool (just like thread on a sewing machine bobbin).
That time the problem was solved by pulling the cable back and forth using the simultaneous power of both the backup and primary motors.

But even that's only half the trouble. Even if we haven't flown too far from our parent star, catching its rays isn't always possible.
Earth satellites, for example, can be kept in a so-called sun-synchronous orbit in the terminator plane (that is, right on the sunset/sunrise line) — then the spacecraft will be constantly illuminated by sunlight and its batteries accordingly.
But what if we need a different orbit? At some point the planet will block the satellite from sunlight — and then all hope rests on batteries. And they, unfortunately, degrade over time.
And solar panels, by the way, also degrade with age — even on Earth they gradually tire from ultraviolet radiation, though very slowly, while in space they also suffer from cosmic radiation and solar flares.
Panels can also be damaged by micrometeorites or space debris, if you're really unlucky.

A detailed account of spacecraft solar panels is beyond our topic, but if you're interested — I recommend a good article (with pictures, too).
So what, what will come to our rescue? Yes, you guessed it.
You remember what's great about an RTG, right? Reliability.
It works for decades without maintenance — and that's exactly what's needed in space.
This is where plutonium-238 comes in handy — its high price turns out to be not so high within the budget of a spacecraft.
Alright, that's an oversimplification — actually, it's still very expensive.

RTGs were successfully used on many satellites and space probes — for example, on Cassini-Huygens and on New Horizons.

And the Voyager probes (both the first and the second) each carried three RTGs with a total power output of 470 watts. And they flew far, far away.

Very far — 25 billion kilometers from home! What other power source can boast that? Exactly.
By the way, here you can track how far from us both Voyagers have flown, in real time — just in case you're curious: NASA Voyager tracker

And in 1971, the SNAP-27 RTG visited the Moon with the Apollo 14 mission.

Improved versions of these generators are also installed on Mars rovers (for example, Curiosity and Perseverance — the solar panels on these vehicles don't provide enough energy during dust storms, which could jeopardize the entire mission. And here, too, reliability is what's needed.

Well okay, things weren't entirely smooth here either (And where are they ever?) — you won't believe it, but those pesky eco-activists came to spoil our fun again.
Actually... well, no one's perfect. There was one very unpleasant incident with launching an RTG into space — and it only took one time, as they say...
On April 21, 1964, during the launch of a rocket carrying the Transit 5BN-3 navigation satellite, an accident occurred. The satellite broke apart and burned up at an altitude of about fifty kilometers somewhere north of Madagascar.
Inside the satellite was an RTG SNAP-9A containing just under a kilogram of plutonium-238. And of course it was pulverized into dust and scattered across the atmosphere. A certain increase in radiation background was detected by measuring stations around the entire Earth. Disaster.
This incident would become a serious argument against launching RTGs into space for many years. However, they didn't actually stop using them — solar panels are fine, of course, but if they don't suit us, then what? Not fly into deep space at all? Boring!

But incidentally, Transit (also known as NAVSAT, from Navy Navigation Satellite System), which lost one of its satellites under such tragic circumstances — was the world's first satellite navigation system. Yes, it didn't cover the entire Earth, and access time was limited — but it worked from the mid-sixties until the mid-nineties!
A lifespan worthy of an RTG. No, not like that. A lifespan worthy — RTG!
By the way, amateur radio operators still pick up signals from these satellites more than half a century after their launch. Isn't that wonderful?
Modern RTGs for the space industry contain nuclear fuel in a capsule capable of surviving even a launch vehicle explosion during a failed launch — engineers know how to learn from their mistakes, you know.
And still, public skepticism has to be taken into account — radiophobia is sometimes widespread even among decision-makers.
But one way or another, in space the RTG apparently has a big future ahead — because the farther from the Sun a spacecraft travels, the less hope there is for solar panels. Moreover, such power sources will be useful at lunar bases when we grow into them, or even on Mars — at a minimum, as a reliable backup.
An energy source lasting for decades — that's great, and fortunately, there are no space looters on the horizon yet. At least we're lucky somewhere!
Back Down to Earth
Now let's come back down to earth. And look at this topic from yet another angle — not the romantic space angle, but the practical terrestrial one.
Recently there's been growing discussion about distributed energy systems with an emphasis on decentralization and distributed generation. One of the promising technologies is small-scale nuclear power plants that can be installed somewhere in remote, hard-to-reach areas, close to the consumers.

The IAEA presented more than fifty small nuclear reactor projects in a 2018 brochure (available online).

But the low efficiency of RTGs remains an obstacle. At NASA, work was being done on an RTG with a Stirling engine (ASRG — Advanced Stirling Radioisotope Generator), in which gas heated by the heat source would drive a piston, which would turn an alternating current generator with an efficiency of 25-30%.

The downside is obvious: moving parts require maintenance. The program was suspended in 2013 due to budget overruns.
We shouldn't expect a significant efficiency increase given the physics of the process. The high cost of nuclear fuel and safety issues at remote sites complicate terrestrial applications. Space remains the more promising direction.

But the work of physicists was not in vain — thermogenerators are used both on Earth and beyond its bounds. The reliability of RTGs has ensured the long-lasting operation of navigation equipment and space missions. The future may bring more efficient solutions and broader use of atomic energy in distributed systems.
In Conclusion (With a Few Sources)
The plan was a short note about RTGs and distant lighthouses. During writing, the text grew unexpectedly.
Recommended reading:
- "Thermoelectricity" by A.S. Bernstein (1957) — fifty pages, excellent writing
- "Compact Nuclear Electrical Power Sources" by Yu.V. Lazarenko, A.A. Pustovalov, V.P. Shapovalov (1992) — a serious work with a 129-entry bibliography, available at Rosatom History
- "Semiconductors in Modern Physics" by Ioffe (1954)
Many illustrations were taken from the IAEA presentation "Prospects for Completing the Russian RTG Disposal Program" (93 slides with photographs, available via the Wayback Machine).